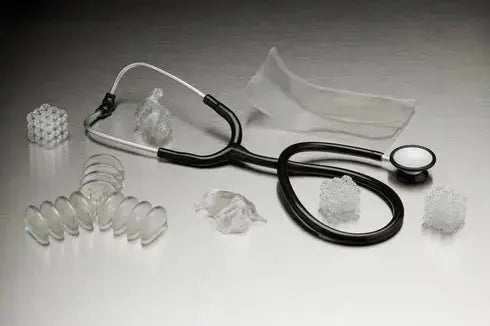
Formlabs BioMed Elastic 50A Resin (Form 3) - 1L is an ISO 10993 and USP Class VI certified medical-grade material designed for applications requiring flexibility and biocompatibility. This soft, translucent material is specifically engineered for the Form 3 series printers, enabling the production of comfortable medical device components and complex anatomical models. Consequently, clinicians can create patient-specific guides and models that simulate the tactile response of soft human tissue whilst maintaining strict regulatory compliance.

Clinical Applications for Formlabs BioMed Elastic 50A Resin (Form 3) - 1L
The material properties of this resin make it indispensable for long-term skin contact and short-term mucosal membrane applications. It is frequently utilised for prototyping respiratory components, patient-specific masks, and cardiovascular models. Furthermore, its 50A Shore durometer ensures that printed parts can withstand significant deformation and return to their original shape, which is critical for functional testing in medical environments.

Integrating Formlabs BioMed Elastic 50A Resin (Form 3) - 1L into a healthcare workflow allows for rapid iteration of medical instruments without the overhead of traditional silicone tooling. This material is manufactured in an ISO 13485 certified facility, ensuring that every litre meets the rigorous quality standards required for clinical use. Using this resin ensures that prototypes are not only functional but also safe for human interaction.

For successful implementation, parts must be post-processed according to Formlabs' validated medical protocols to ensure biocompatibility is maintained. This includes specific wash durations in Isopropyl Alcohol and targeted post-cure cycles. Failure to follow these steps may compromise the material's certified properties, whilst adhering to them guarantees a professional medical-grade finish for every print.